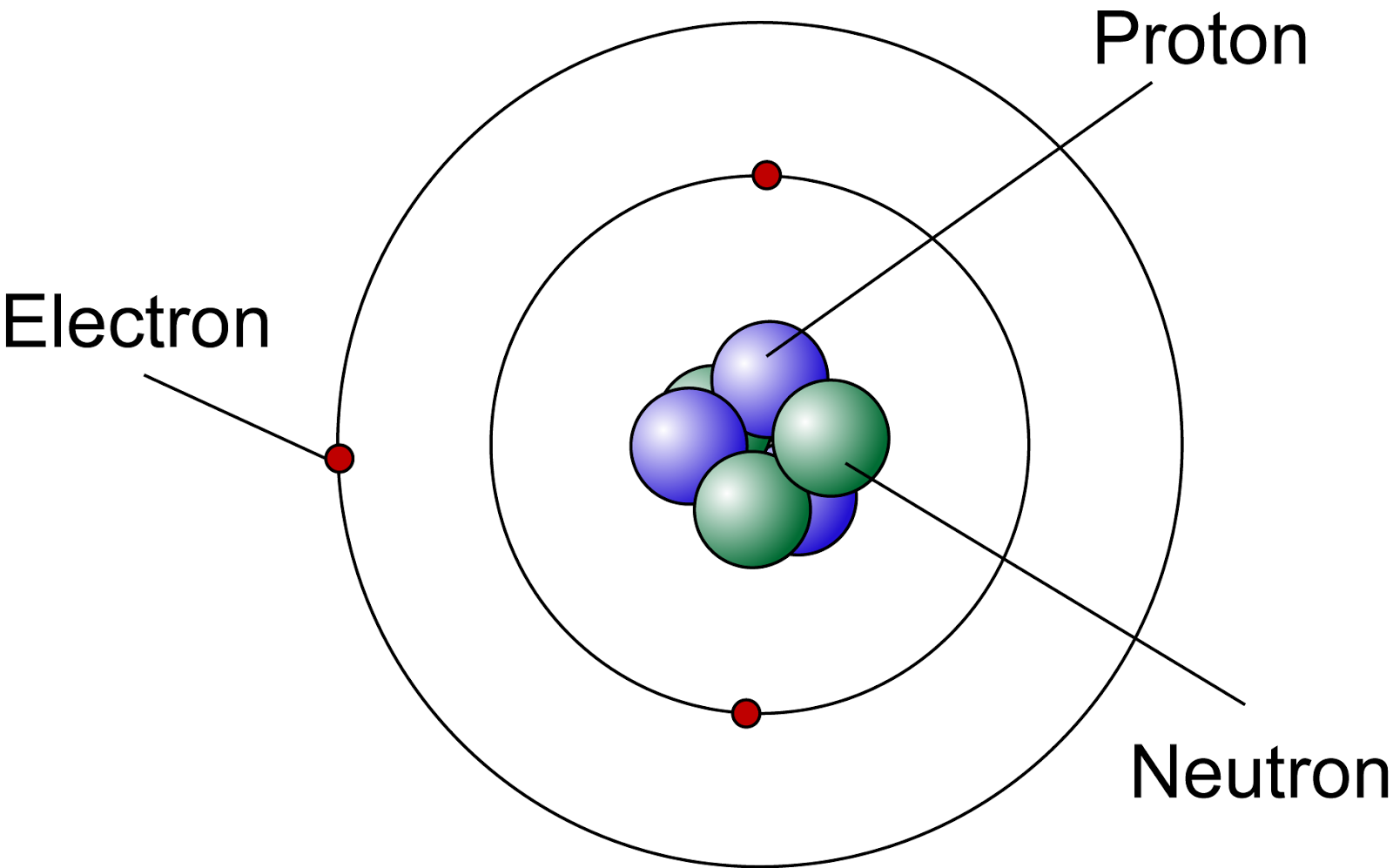
Generally, classical physics is often used to explain physical phenomena at a macroscopic level. The theory basically explains the nature and behaviour of matter and energy on the atomic level. We also acknowledge previous National Science Foundation support under grant numbers 1246120, 1525057, and. Quantum theory is the foundational theory for explaining subatomic phenomena. the California State University Affordable Learning Solutions Program, and Merlot. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. 
A possible pair of compounds that would fit this relationship would be A CO2 and B CO. 
This means that A and B are different compounds, with A having one-half as much carbon per amount of oxygen (or twice as much oxygen per amount of carbon) as B. Trending Questions How man grams in A9 tin? How did Daniel Bernoulli contribute to the particle theory? Isotopes are the atoms of same element having same but different atomic massA mass number B number of neutions C atomic numberD number of electrons? Which one of the following is an example of a strong acid? What is the rarest naturally occurring element? What is MnI4? What kinds of substanances dissolve in water? What is the process turning gas to liquid? A chemical formula is an expression used to represent? What would break down sweetcorn fibers to make paper? How many electrons neutrons and protons are in the nucleus of einsteinium? A substance that helps to keep a stable pH in a solution is called a? How does vinegar have an alkaline effect on the body Need explanation in words.? What is Case TCH fluids? What is the number of potassium atoms compared to oxygen atoms compared to oxygen atoms in a binary compound made from these two elements? How many electrons would make the d shell complete? What processes change water to vapor in air and back to iquid water? If you make up a solution of 100 mL of 0. Explore an atoms interior to discover the layout of its nucleus, protons, and electrons. State the postulates of Dalton’s atomic theory Use postulates of Dalton’s atomic theory to explain the laws of definite and multiple proportions. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. This supports the law of multiple proportions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
